You can check 50+ pages 5.0 liters of a 0.1 m ca oh 2 solution explanation in PDF format. What volume of 126 M HCl must be added to enough water to prepare 500 liters of 300 M HCl. Therefore because 2 moles of H Cl react for every 1 mole of CaOH2 that reacts 0337 2 0674 moles of H Cl will react. How many moles of KI potassium iodide are present in 125 ml of 05 M KI. Check also: solution and 5.0 liters of a 0.1 m ca oh 2 solution 3250 103 M 310 107 M 420 1012 M correct 510 M Explanation.
A 05 M solution. G d 64 g e none of these ANS.
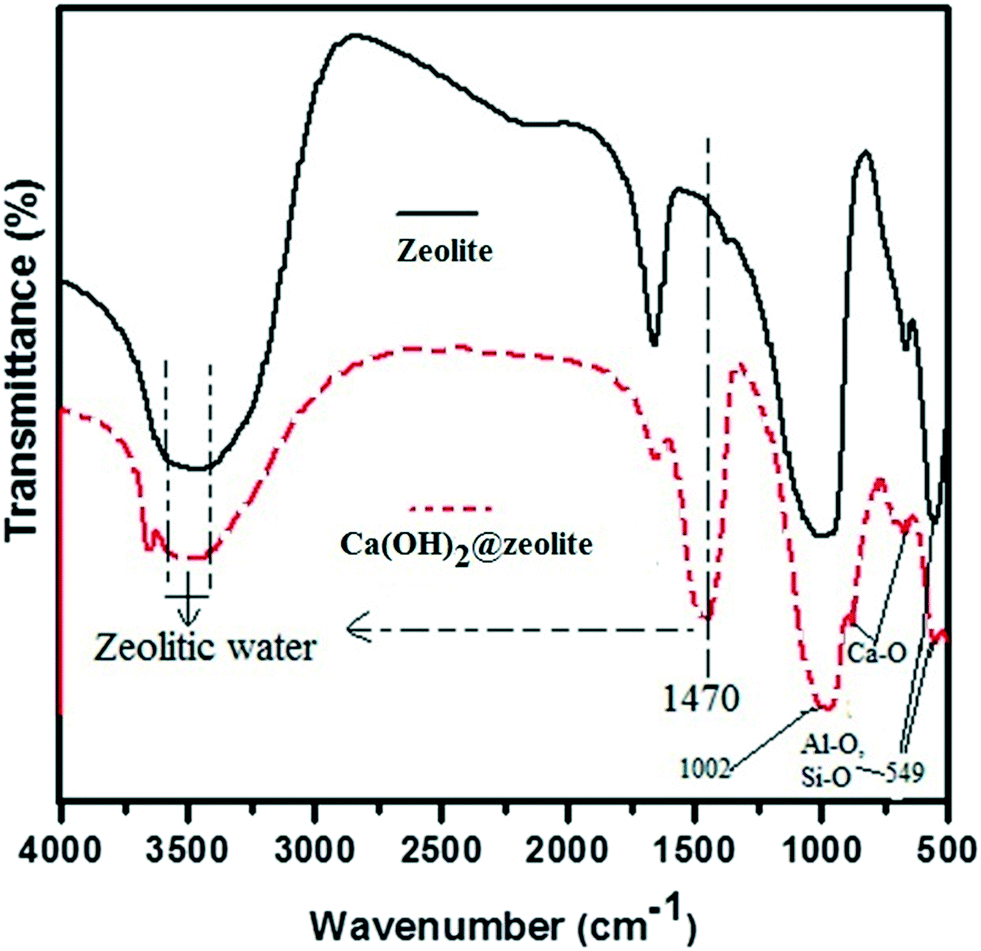
A Novel Nanoposite Of Ca Oh 2 Incorporated Zeolite As An Additive To Reduce Atmospheric Emissions Of Pm And Vocs During Asphalt Production Environmental Science Nano Rsc Publishing Doi 10 1039 C6en00483k 12 g of lithium hydroxide LiOH in 10 L of solution.
| Topic: Calculate the molarity of the following solutions. A Novel Nanoposite Of Ca Oh 2 Incorporated Zeolite As An Additive To Reduce Atmospheric Emissions Of Pm And Vocs During Asphalt Production Environmental Science Nano Rsc Publishing Doi 10 1039 C6en00483k 5.0 Liters Of A 0.1 M Ca Oh 2 Solution |
| Content: Learning Guide |
| File Format: DOC |
| File size: 3mb |
| Number of Pages: 24+ pages |
| Publication Date: January 2018 |
| Open A Novel Nanoposite Of Ca Oh 2 Incorporated Zeolite As An Additive To Reduce Atmospheric Emissions Of Pm And Vocs During Asphalt Production Environmental Science Nano Rsc Publishing Doi 10 1039 C6en00483k |
 |
Calculate the molarity of the following solutions.

First find the number of moles of KCl in the 2500 mL of 0500 M solution. 110 L of 05 M CaOH 2 3a. Calculate the mass of solute needed to prepare each of the following solutions. 12 g of lithium hydroxide LiOH in 10 L of solution b. A 20 L of 18 M NaOH solution. Calculate the number of moles and the number of grams of solute in each solution.

Calcium Hydroxide Doped Kno 3 As A Promising Candidate For Thermochemical Storage Of Solar Heat Rsc Advances Rsc Publishing Doi 10 1039 C7ra06639b NH 4 2 SO 3 produces three ions for every one formula unit that dissolves.
| Topic: 27045 L x 0090 mol 1 L b 20 L of 15 M NaOH solution. Calcium Hydroxide Doped Kno 3 As A Promising Candidate For Thermochemical Storage Of Solar Heat Rsc Advances Rsc Publishing Doi 10 1039 C7ra06639b 5.0 Liters Of A 0.1 M Ca Oh 2 Solution |
| Content: Explanation |
| File Format: DOC |
| File size: 800kb |
| Number of Pages: 7+ pages |
| Publication Date: December 2017 |
| Open Calcium Hydroxide Doped Kno 3 As A Promising Candidate For Thermochemical Storage Of Solar Heat Rsc Advances Rsc Publishing Doi 10 1039 C7ra06639b |
 |

A Novel Nanoposite Of Ca Oh 2 Incorporated Zeolite As An Additive To Reduce Atmospheric Emissions Of Pm And Vocs During Asphalt Production Environmental Science Nano Rsc Publishing Doi 10 1039 C6en00483k 107 g NH 4 Cl 1b.
| Topic: 100 mL of a 05 M NH 4 3 PO 4 solution 2. A Novel Nanoposite Of Ca Oh 2 Incorporated Zeolite As An Additive To Reduce Atmospheric Emissions Of Pm And Vocs During Asphalt Production Environmental Science Nano Rsc Publishing Doi 10 1039 C6en00483k 5.0 Liters Of A 0.1 M Ca Oh 2 Solution |
| Content: Synopsis |
| File Format: Google Sheet |
| File size: 2.1mb |
| Number of Pages: 4+ pages |
| Publication Date: March 2019 |
| Open A Novel Nanoposite Of Ca Oh 2 Incorporated Zeolite As An Additive To Reduce Atmospheric Emissions Of Pm And Vocs During Asphalt Production Environmental Science Nano Rsc Publishing Doi 10 1039 C6en00483k |
 |

How To Write The Ionic Equation For Hcl Ca Oh 2 Cacl2 H2o A 16 g b 80.
| Topic: What volume of 01 M hydrochloric acid will react with 26 grams of zinc. How To Write The Ionic Equation For Hcl Ca Oh 2 Cacl2 H2o 5.0 Liters Of A 0.1 M Ca Oh 2 Solution |
| Content: Synopsis |
| File Format: PDF |
| File size: 1.7mb |
| Number of Pages: 24+ pages |
| Publication Date: July 2018 |
| Open How To Write The Ionic Equation For Hcl Ca Oh 2 Cacl2 H2o |
 |

Pdf Phase Diagram And Volume Change Of The Ca Oh 2 Cacl2 H2o System For Varying Ca Oh 2 Cacl2 Molar Ratios How many liters of hydrogen will you make at STP if you react 274 L of 045 M hydrochloric acid with excess zinc.
| Topic: 1 The chemical equation. Pdf Phase Diagram And Volume Change Of The Ca Oh 2 Cacl2 H2o System For Varying Ca Oh 2 Cacl2 Molar Ratios 5.0 Liters Of A 0.1 M Ca Oh 2 Solution |
| Content: Answer |
| File Format: PDF |
| File size: 1.5mb |
| Number of Pages: 21+ pages |
| Publication Date: February 2021 |
| Open Pdf Phase Diagram And Volume Change Of The Ca Oh 2 Cacl2 H2o System For Varying Ca Oh 2 Cacl2 Molar Ratios |
 |
Calcium Hydroxide Doped Kno 3 As A Promising Candidate For Thermochemical Storage Of Solar Heat Rsc Advances Rsc Publishing Doi 10 1039 C7ra06639b A 10 M solution containing 85 g of.
| Topic: A Show a correct numerical setup for calculating the total number of moles of CaNO32 needed to make 0250 liter of the 0200 M calcium nitrate solution. Calcium Hydroxide Doped Kno 3 As A Promising Candidate For Thermochemical Storage Of Solar Heat Rsc Advances Rsc Publishing Doi 10 1039 C7ra06639b 5.0 Liters Of A 0.1 M Ca Oh 2 Solution |
| Content: Solution |
| File Format: DOC |
| File size: 810kb |
| Number of Pages: 9+ pages |
| Publication Date: July 2021 |
| Open Calcium Hydroxide Doped Kno 3 As A Promising Candidate For Thermochemical Storage Of Solar Heat Rsc Advances Rsc Publishing Doi 10 1039 C7ra06639b |
 |

Balance This Equation Properly Explaining It In Table Hno3 Ca Oh 2 Gt Ca No3 2 H2o Brainly In 100 mL of a 05 M NH43PO4 solution.
| Topic: 230745 mol 0415 liters 179 moles liter M 2. Balance This Equation Properly Explaining It In Table Hno3 Ca Oh 2 Gt Ca No3 2 H2o Brainly In 5.0 Liters Of A 0.1 M Ca Oh 2 Solution |
| Content: Synopsis |
| File Format: PDF |
| File size: 5mb |
| Number of Pages: 27+ pages |
| Publication Date: February 2020 |
| Open Balance This Equation Properly Explaining It In Table Hno3 Ca Oh 2 Gt Ca No3 2 H2o Brainly In |
 |

S Pubs Acs Doi Pdf 10 1021 Acs Iecr 5b02356 What mass of the following chemicals is needed to make the solutions indicated.
| Topic: HCOOH H HCOO weak acid. S Pubs Acs Doi Pdf 10 1021 Acs Iecr 5b02356 5.0 Liters Of A 0.1 M Ca Oh 2 Solution |
| Content: Analysis |
| File Format: Google Sheet |
| File size: 1.7mb |
| Number of Pages: 9+ pages |
| Publication Date: October 2020 |
| Open S Pubs Acs Doi Pdf 10 1021 Acs Iecr 5b02356 |
 |

A Prehensive Study On The Dominant Formation Of The Dissolved Ca Oh 2 Aq In Strongly Alkaline Solutions Saturated Ca Ii Rsc Advances Rsc Publishing Doi 10 1039 C6ra05337h 15 mol 20 L x 30 mol 1 L 8.
| Topic: 100 mL of a 05 M NH 4 3 PO 4 solution 2. A Prehensive Study On The Dominant Formation Of The Dissolved Ca Oh 2 Aq In Strongly Alkaline Solutions Saturated Ca Ii Rsc Advances Rsc Publishing Doi 10 1039 C6ra05337h 5.0 Liters Of A 0.1 M Ca Oh 2 Solution |
| Content: Synopsis |
| File Format: DOC |
| File size: 2.6mb |
| Number of Pages: 17+ pages |
| Publication Date: April 2020 |
| Open A Prehensive Study On The Dominant Formation Of The Dissolved Ca Oh 2 Aq In Strongly Alkaline Solutions Saturated Ca Ii Rsc Advances Rsc Publishing Doi 10 1039 C6ra05337h |
 |

How Many Atoms Are Contained In A Mole Of Ca Oh 2 Once equilibrated some solid CaOH 2 remains undissolved.
| Topic: X 0615 L 121 g 1161392 gmol x 0169407 M --- Ill carry some guard digits calculate the concentration of all ions. How Many Atoms Are Contained In A Mole Of Ca Oh 2 5.0 Liters Of A 0.1 M Ca Oh 2 Solution |
| Content: Answer |
| File Format: DOC |
| File size: 1.9mb |
| Number of Pages: 30+ pages |
| Publication Date: March 2019 |
| Open How Many Atoms Are Contained In A Mole Of Ca Oh 2 |
 |

Nanomaterials Free Full Text Halloysite Nanotubes Controlled Access And Release Smart Gates Html A 16 g PAGE.
| Topic: The following morning the solution is 110 M. Nanomaterials Free Full Text Halloysite Nanotubes Controlled Access And Release Smart Gates Html 5.0 Liters Of A 0.1 M Ca Oh 2 Solution |
| Content: Summary |
| File Format: Google Sheet |
| File size: 1.8mb |
| Number of Pages: 21+ pages |
| Publication Date: March 2021 |
| Open Nanomaterials Free Full Text Halloysite Nanotubes Controlled Access And Release Smart Gates Html |
 |

A Novel Nanoposite Of Ca Oh 2 Incorporated Zeolite As An Additive To Reduce Atmospheric Emissions Of Pm And Vocs During Asphalt Production Environmental Science Nano Rsc Publishing Doi 10 1039 C6en00483k 3 the ammonium sulfite solution.
| Topic: What volume of solvent has evaporated from the 0275 M solution. A Novel Nanoposite Of Ca Oh 2 Incorporated Zeolite As An Additive To Reduce Atmospheric Emissions Of Pm And Vocs During Asphalt Production Environmental Science Nano Rsc Publishing Doi 10 1039 C6en00483k 5.0 Liters Of A 0.1 M Ca Oh 2 Solution |
| Content: Answer |
| File Format: DOC |
| File size: 1.9mb |
| Number of Pages: 23+ pages |
| Publication Date: August 2021 |
| Open A Novel Nanoposite Of Ca Oh 2 Incorporated Zeolite As An Additive To Reduce Atmospheric Emissions Of Pm And Vocs During Asphalt Production Environmental Science Nano Rsc Publishing Doi 10 1039 C6en00483k |
 |
First find the number of moles of KCl in the 2500 mL of 0500 M solution. What is the final pH of this buffer assuming K a formic acid 180 x 10 4. Write out the reaction of the acid dissociation.
Its really simple to prepare for 5.0 liters of a 0.1 m ca oh 2 solution 198 g of barium bromide BaBr2 in 20 L of solution. A 20 L of 18 M NaOH solution. 110 L of 05 M CaOH 2 3a. A prehensive study on the dominant formation of the dissolved ca oh 2 aq in strongly alkaline solutions saturated ca ii rsc advances rsc publishing doi 10 1039 c6ra05337h a prehensive study on the dominant formation of the dissolved ca oh 2 aq in strongly alkaline solutions saturated ca ii rsc advances rsc publishing doi 10 1039 c6ra05337h pdf phase diagram and volume change of the ca oh 2 cacl2 h2o system for varying ca oh 2 cacl2 molar ratios how to write the ionic equation for hcl ca oh 2 cacl2 h2o calcium hydroxide doped kno 3 as a promising candidate for thermochemical storage of solar heat rsc advances rsc publishing doi 10 1039 c7ra06639b calcium hydroxide doped kno 3 as a promising candidate for thermochemical storage of solar heat rsc advances rsc publishing doi 10 1039 c7ra06639b a novel nanoposite of ca oh 2 incorporated zeolite as an additive to reduce atmospheric emissions of pm and vocs during asphalt production environmental science nano rsc publishing doi 10 1039 c6en00483k how many moles of ca oh 2 are needed to neutralize th 2325 Ca 2 O 2 H 25 4008 2 1600 2 1008.